C4.1: Site-Selective Coupling to Electrodes and Transport through Single Molecules
Subproject Leader: Hilbert v. Löhneysen
Contributing Scientists:
Present: Ralph Krupke, Maya Lukas, Manfred Kappes, Cornelius Thiele, Marcel Mayor, Christophe Stroh, Frank Hennrich
Past: Christoph Marquardt, Kerrin Dössel, Alexandrina Stuparu, Carmen Pérez León, Tihomir Tomanic, Christoph Sürgers
Carbon Nanotubes as Molecular Leads
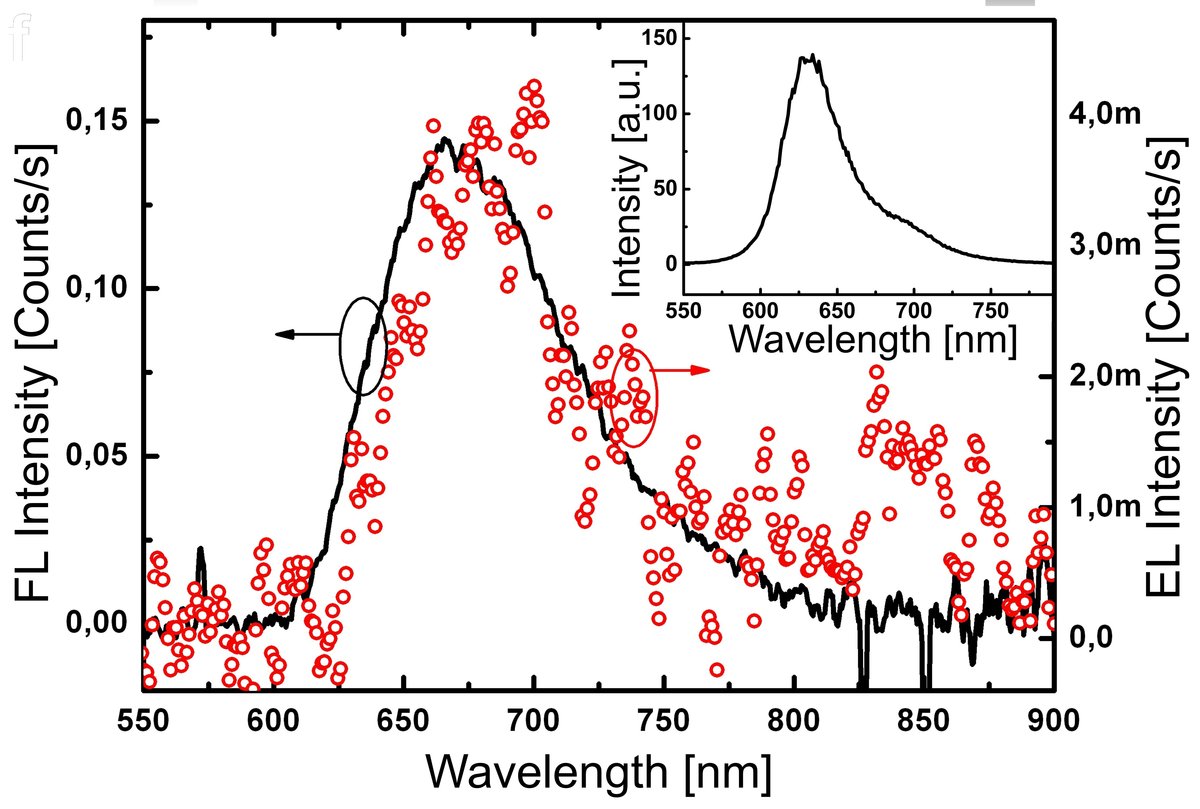
In this subproject we investigate electron transport through tailored single molecules. Carbon nanotubes could be ideal molecular leads to contact small molecules, provided a tiny gap can be opened that can accommodate single molecules. In a first approach, defect formation in carbon nanotubes induced by electron irradiation was studied [1]. We discovered a transition from the low-resistance metallic to the high-resistance insulating state, which surprisingly could be reversed by a large voltage bias. This cyclic process, which can be repeated, is not due to an opening and closing of a gap, but due to charges that are trapped in and released from the substrate. Successful gap formation was achieved by a controlled electric breakdown of free-standing nanotube segments [2]. Gaps with a dimension < 10 nm could be formed at low oxygen partial pressure. Into such gaps we succeeded to position an elongated specifically tailored organic chromophore molecule, and observed electroluminescence from a single molecule (“single-molecule flashlight”). The tailored molecules are synthesized in subproject C3.8.
Three-Dimensional Molecules
Single wire-like molecules usually bind with their long axis parallel to metallic electrodes: Using a scanning tunneling microscope (STM) we thus face the problem of measuring the conductance of a bare wire that lies on a metal table plate. Lifting this wire at one side with the STM will change the bond to the surface at the other side and the conformation of the molecule, leaving us with a set of unknown parameters that all have an influence on the conductance. We therefore investigated a rigid tripodal molecule with three molecular wires (“legs”) connected to a metal complex (“head”). The majority of the molecules were found with two legs and the head on the Au(111) surface and the third leg protruding into vacuum. This allowed us to position the STM tip above different parts of the molecule, and investigate the electronic structure and conductance of the protruding (insolated) wire as compared to the wire that lies on the surface (bare, short circuited wire) [3]
Macromolecules: a Bottom-Up Approach
In a preliminary work to deposit large p-conjugated macrocycles on metal surfaces, we explored the possibility to deposit segments of these macrocycles, e.g., C114H158O8S2Si2 (CHOSSi) by pulse injection with THF solvent molecules on Cu(111). We were able to image the solvent besides the CHOSSi molecules at room temperature, indicating that the solvent is immobilized by the latter and, in turn, that the CHOSSi molecules are deposited as individual species without clustering [4].
References
| [1] | C.W. Marquardt, S. Dehm, A. Vijayaraghavan, S. Blatt, F. Hennrich, and R. Krupke, Nanotubes, Nano Lett. 9, 2767 (2008) |
| [2] | C.W. Marquardt, S. Grunder, A. Błaszczyk, S. Dehm, F. Hennrich, H. v. Löhneysen, M. Mayor, and R. Krupke, Nature Nanotechnology 5, 863 (2010) |
| [3] | K. Dössel, M. Lukas, A. Schramm, K. Fink, O. Fuhr, C. Stroh, M. Mayor, and H. v. Löhneysen, submitted for publication |
| [4] | C. Pérez León, C. Sürgers, M. Mayor, M. Marz, R. Hoffmann, and H. v. Löhneysen, J. Phys. Chem. C 113, 14335 (2009) |
List of Publications 2006-2011 as PDF
Subproject Report 2006-2010 as PDF