C1.2: Synthesis and Characterization of New Nano-Scale Aggregates Displaying Cooperative Magnetic Coupling
Subproject Leader: Annie Powell
Institut für Nanotechnologie, KIT
Contributing Scientists:
Present: Manase Ako Ayuk, Yanhua Lan
Single Molecule Magnets
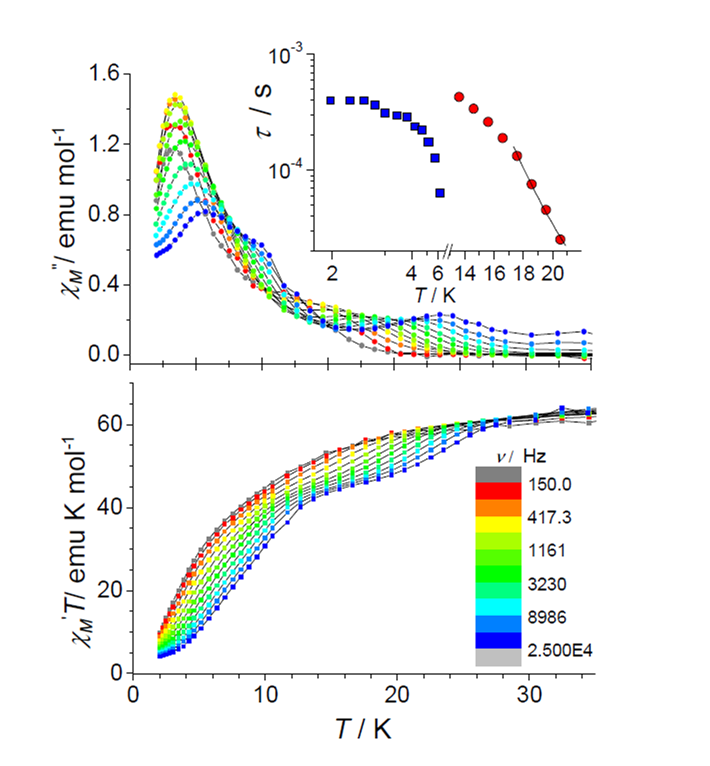
Single-Molecule Magnets (SMMs) are molecular coordination clusters of paramagnetic metal cations that behave as individual nanoscale magnets, which show magnetic hysteresis, and in some cases also Quantum Tunneling of Magnetisation (QTM). These have obvious potential applications in high-density data storage and quantum computing.
Although the first SMM was identified in 1991, surprising little progress has been made since in optimising their properties, with blocking temperatures (the temperature below which they can retain their magnetisation for a significant length of time) still generally below 4 K. The height of the energy barrier Ueff to the reorientation of the ground state spin S is given by Ueff = -DS2 (for an integer spin S). To obtain SMMs with higher, more useful blocking temperatures (above 4 K; as a longer term goal, above 78 K), we need to increase the height of this barrier by combining a large spin S and a negative (i.e. uniaxial) anisotropy D within the same molecule, and our main area of research currently involves meeting this great synthetic challenge.
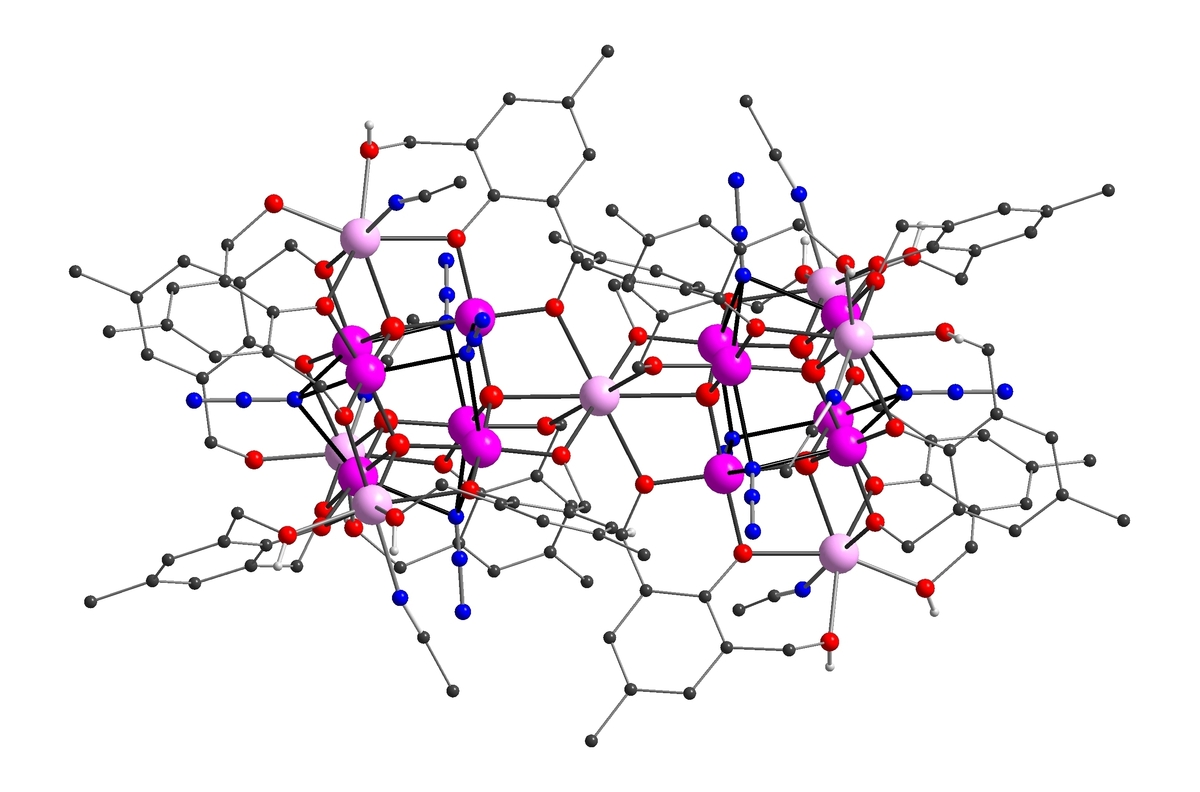
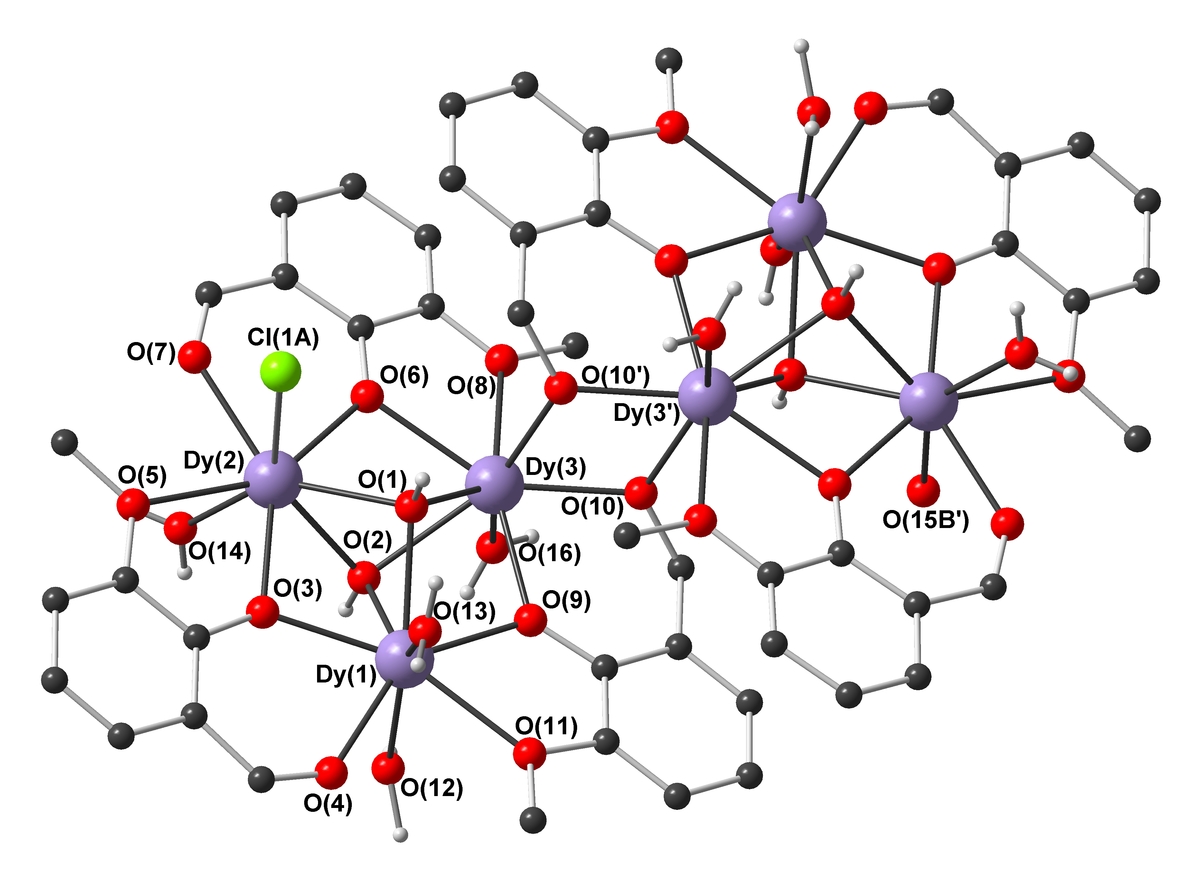
We have synthesized a Mn19 coordination cluster which still holds the record ground state spin: S = 83/2. Ferromagnetic interactions align all the 83 unpaired electrons in the molecule parallel[1]. By selectively replacing the isotropic MnII ion at the centre of the molecule with the anisotropic DyIII ion, SMM behaviour could be induced. However, we are now concentrating on optimizing the Ising-type anisotropy D, and have synthesised a range of SMMs incorporating highly anisotropic metal ions (e.g. DyIII6, MnIVMnIII4DyIII4), with high Ueff barriers and complicated magnetic slow relaxation behavior [2,3].
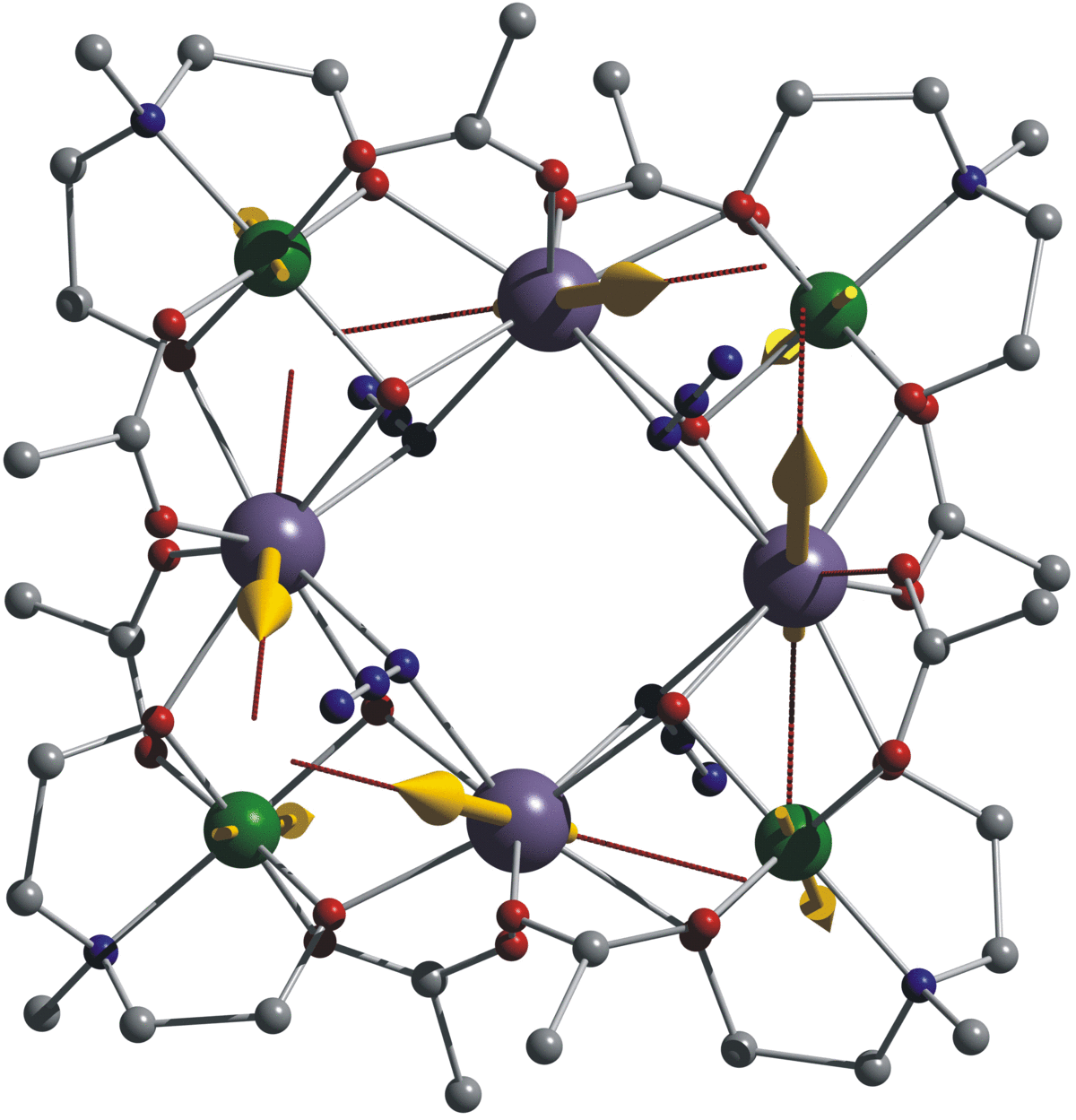
Understanding and Controlling Magnetic Anisotropy
The synthesis of coordination clusters containing highly anisotropic metal ions is one thing, but aligning their single-ion anisotropies so as to obtain a large negative molecular D is a severe synthetic and theoretical challenge – often the individual D tensors cancel each other out within the molecule. Quantum-chemical CASSCF/CASPT2/RASSI-SO calculations show how the coordination geometry of an ion directs the orientation of its single-ion anisotropy tensor, and how these then combine to give the molecular anisotropy D. This overall D can then be related to the observed slow relaxation of the magnetization[4]. These findings can then inform our synthetic strategy.
57Fe Mößbauer Spectroscopy as a Probe of Spin State and Anisotropy
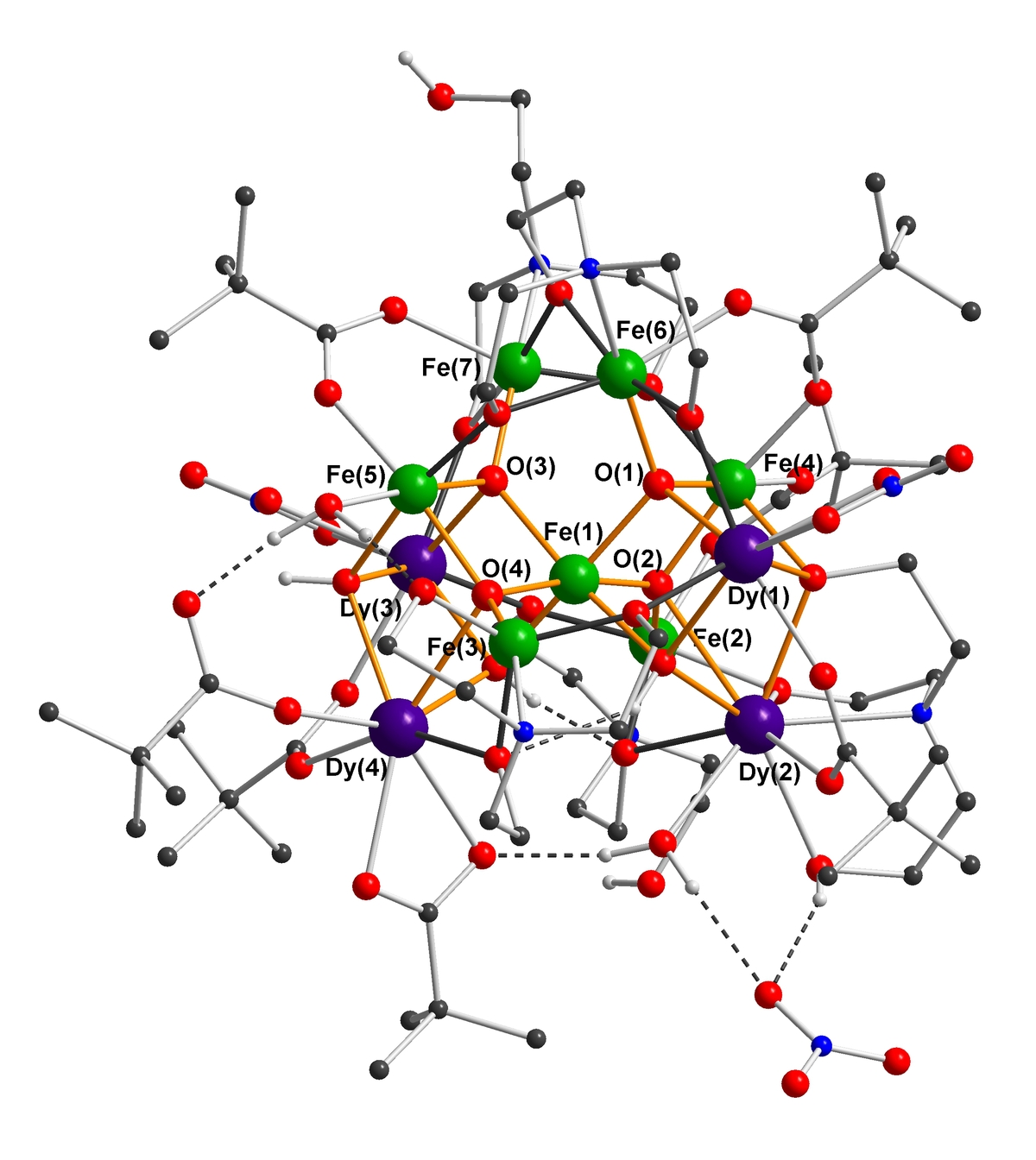
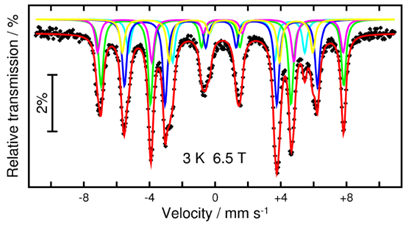
We have shown that 57Fe Mößbauer spectra measured under a range of magnetic fields can give maps of the FeIII spins in large clusters (up to Fe16) and can give such information even in the presence of highly anisotropic lanthanide ions (e.g. in a Fe7Dy4 cluster) [5]. Mößbauer spectroscopy can measure the local hyperfine field “felt” by Fe nuclei in a molecule. In heterometallic Fe-4f clusters such fields derive mainly from the magnetic anisotropies of the lanthanide cations, and our goal is to derive a methodology to obtain information from the Mößbauer spectra on the orientations of their single-ion anisotropies relative to the molecular structure. Such new applications of Mößbauer spectroscopy give information complementary to that obtainable from SQUID susceptibility measurements.
Super Metal-Organic Frameworks (SMOFs)
In the so-called Metal-Organic Frameworks (MOFs), relatively small (generally up to tetranuclear) metal complex nodes are generally linked by long polyaromatic organic ligands to build up highly-porous materials. We are now adopting the converse approach, building large high-nuclearity clusters into 3-D networks via rather small bridging ligands. Such networks, which we term Super Metal-Organic Frameworks or SMOFs, can still be highly porous, but the cavities now have a significant contribution from the surface of the aggregates themselves, allowing a variety of functionalities to be available to a guest molecule.
We have found that such SMOFs can combine SMM behavior with efficient catalytic activity. Other properties such as luminescence and gas storage are under investigation, and such SMOFs are potentially true multifunctional materials. We also find that they can be pyrolysed to give nanostrucutred metal oxide materials.
References
|
[1] |
A.M. Ako, I.J. Hewitt, V. Mereacre, R. Clérac, W. Wernsdorfer, C.E. Anson and A.K. Powell, Angew. Chem. Int. Ed., 45, 4296-4299 (2006) |
|
[2] |
V. Mereacre, A.M. Ako, R. Clérac, W. Wernsdorfer, I.J. Hewitt, C.E. Anson, and A.K. Powell, Chem. Eur. J., 14, 3577-3584 (2008) |
|
[3] |
I.J. Hewitt, J. Tang, N.T. Madhu, C.E. Anson, Y. Lan, J. Luzon, M. Etienne, R. Sessoli and A.K. Powell, Angew. Chem. Int. Ed., 49, 6352-6356 (2010) |
|
[4] |
J. Rinck, G. Novitchi, W. Van den Heuvel, L. Ungur, Y. Lan, W. Wernsdorfer, C.E. Anson, L.F. Chibotaru and A.K. Powell, Angew. Chem. Int. Ed., 49, 7583-7587 (2010) |
|
[5] |
V. Mereacre, D. Prodius, Y. Lan, C. Turta, C.E. Anson and A.K. Powell, Chem., Eur. J., 17, 123-128 (2011) |
List of Publications 2006-2011 as PDF
Subproject Report 2006-2010 as PDF