F1.1: New Concepts for Hybrid Solar Cells
Subproject Leader: Vorname Name
| Heinz Kalt | Institut für Angewandte Physik, KIT |
| Uli Lemmer | Lichttechnisches Institut, KIT |
Contributing Scientists:
Present: Jonas Conradt, Zhenhao Zhang
Past: Daniel Rülke, Torsten Beck, Matthias Karl, Nico Christ, Hung Thai Do, Jan Brückner, Manuel Reinhard, Klaus Huska
In subproject F1.1, new concepts for efficient and low-cost solar cells are investigated. In order to enable efficient charge-carrier extraction in polymer solar cells typically a p-i-n architecture is utilized. While there are several organic p-doped semiconductors that can be deposited from solution, there is still a lack of suitable n-doped counterparts. Hence, the introduction of the inorganic n-type semiconductor zinc oxide (ZnO) has been proven to be advantageous since it can be easily n-doped by various metals.
In this project, arrays of vertically aligned, crystalline ZnO nanorods are integrated into organic solar cells. The nanorods can then serve as efficient pathways for electron extraction from the organic absorber blend. Furthermore, layers of doped ZnO nanoparticles are implemented as charge transport layers in organic single and tandem solar cells
Zinc Oxide Nanorods for Hybrid Solar Cells
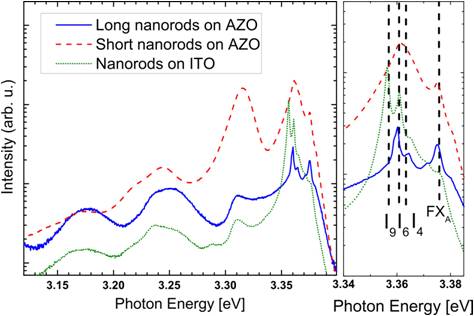
ZnO nanorod arrays were grown using a vapor phase transport method in a horizontal tube furnace [1]. This growth method does not require a catalyst, works at 510°C and allows for the growth of highly crystalline, well-aligned ZnO nanorod arrays. The nanorod length is tunable, ranging from hundreds of nanometers to several micrometers. In addition, the grown nanorods exhibit an excellent crystal quality, as shown by photoluminescence measurements and high-resolution transmission electron microscopy [2, 3].
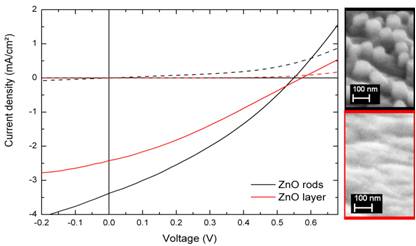
In order to incorporate the ZnO nanorod arrays on AZO substrates into hybrid solar cells, we fabricate devices in the architecture AZO / ZnO / P3HT:PCBM / PEDOT:PSS / Al and compared two different shapes of nanostructures. While the nanorod array exhibits a maximum height of approximately 200nm we also utilized rather smooth ZnO layers for comparison. The open circuit voltage VOC of the device comprising nanorods is slightly lower than the VOC of the cell incorporating a plane ZnO layer which we attribute to possible shunt paths through long ZnO rods. On the other hand, the short circuit current is significantly increased by the introduction of the nanorod array indicating enhanced electron extraction out of the organic blend and hence allowing for an increased efficiency of the hybrid photovoltaic device.
Indium-Doped Zinc Oxide Nanoparticles for Hybrid Solar Cells
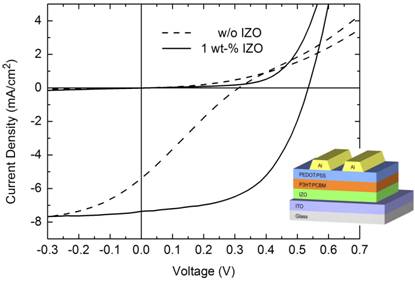
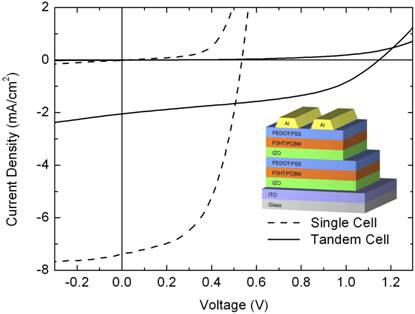
Additionally, inverted organic solar cells using compact layers of indium-doped ZnO (IZO) nanoparticles as inorganic n-doped semiconductor material are investigated. IZO nanoparticles are synthesized by a method based on microwave-assisted polyol synthesis developed in CFN project F1.3. The incorporation of indium as dopant into the ZnO nanoparticles allows for proper energetic alignment between the absorber blend and the cathode. Hence, we fabricate inverted hybrid solar cells exhibiting energy conversion efficiencies of up to 2.3%. We further present hybrid recombination zones comprising a stack of a PEDOT:PSS layer and an IZO nanoparticle layer allowing for the fabrication of hybrid tandem solar cells. The open circuit voltage of the tandem cell is almost 1.2 V which clearly indicates a proper working monolithic tandem cell [4].
References
|
[1] |
H.J. Zhou, J. Fallert, J. Sartor, R.J.B. Dietz, C. Klingshirn, H. Kalt, D. Weissenberger, D. Gerthsen, H.B. Zeng, W.P. Cai, Appl. Phys. Lett. 92, 132112 (2008) |
|
[2] |
J. Sartor, F. Maier-Flaig, J. Conradt, J. Fallert, H. Kalt, D. Weissenberger, D. Gerthsen, Phys. Status Solidi C 7, 1583 (2010) |
|
[3] |
J. Conradt, J. Sartor, C. Thiele, F. Maier-Flaig, J. Fallert, H. Kalt, R. Schneider, M. Fotouhi, P. Pfundstein, V. Zibat, D. Gerthsen, J. Phys. Chem. C 115, 3539 (2011) |
|
[4] |
A. Puetz, T. Stubhan, M. Reinhard, O. Loesch, E. Hammarberg, S. Wolf, C. Feldmann, H. Kalt, A. Colsmann, U. Lemmer, Sol. En. Mat. & Sol. Cells 95, 579 (2011) |
List of Publications 2006-2011 as PDF
Subproject Report 2006-2010 as PDF