C5.2: Synthesis of Functionalized Organic Nanostructures
Subproject Leader: Stefan Bräse
Institut für Organische Chemie, KIT
Contributing Scientists:
Present: Marion Döbele, Dorota Jacubcyk
Past: Manuel Bröhmer, Oliver Plietzsch, Celine Rethore, Olga Lemke, Stefan Seifermann, Katarzyna Koroniak, Dagmar Kapeller, Philippe Pierrat, Frank Hahn
3D Organic Molecules as Building Blocks for Self-Assembled Nanostructures.
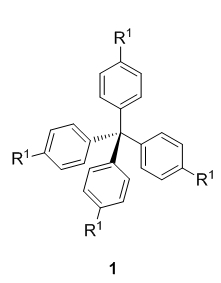
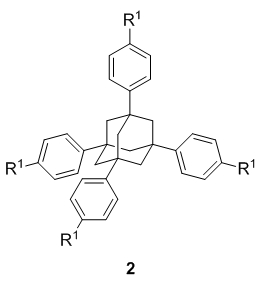
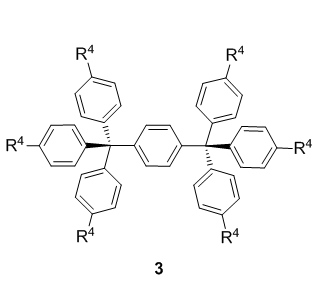
In subproject C5.2, purely organic building cores are designed and prepared on synthetically useful scales in order to serve for the generation of higher molecular assemblies. These rigid three dimensional purely organic molecules comprise methanes 1, 1,3,5,7-tetrasubstitued adamantanes 2, p-bis(triphenylmethyl)benzene derivatives 3 and hexakis fullerene adducts 4 (Figure 1).
All core structures 1–4 are meant to be easily functionalizable at the R-positions in order to introduce various anchor groups, so-called sticky sites that allow network formation via self-assembly.
In this context, we have developed two general approaches starting from common precursors of 1 and 2 and leading to a whole series of different building blocks. The first of these very efficient strategies is based on Click chemistry (1) and the second one on organo-metallic cross-coupling reactions (2). Both procedures are high yielding, easy to operate and allow preparing sufficient quantities to generate supramolecular assemblies.
We have furthermore developed the first fourfold asymmetric addition on adequate 1 and 2 type precursors in order to obtain peripheral chiral building block which will later on sever for the generation of enantiomerically pure networks (3).
In the fullerene series 4, a hexakis crown-ether adduct has been prepared that can easily been functionalized at its six end positions (4).
We are about to publish our results on a very efficient access to type 3 precursor molecules.
Self-Assembly of Various Networks.
With our organic building units, we managed together with our collaboration partners, to prepare different networks.
Self-Assembly via H-bonding
In co-operation with Prof. Clemens Richert (Universität Stuttgart), we were able to prepare core structures 1, 2 and 3 bearing self-complementary DNA-strands. Network generation occurred through Watson-Crick base pairing and delivered a novel material with an exceptionally high melting point (5, 6).
Self-Assembly via covalent-bonding
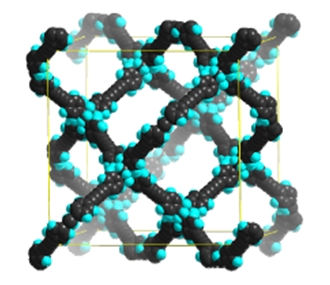
In co-operation with Prof. Dan Zhao (University of Texas), we were able to prepare Porous Organic Frameworks (POFs) via organic-metallic cross-coupling reactions. The resulting networks showed a remarkable selectivity for methane – carbon dioxide mixtures (7) (Figure 2).
Prof. Christopher Barner-Kowollik (KIT) used our hexakis crown-ether fullerene adducts to produce well-defined star shape polymers via the Click reaction (8).
In the same context, co-operating with Dr. Angiolina Comotti and Prof. Piero Sozzani (Università degli Studi di Milano Bicocca), we have very recently been able to generate Hyper Cross-linked Polymers (HCPs) via Click Chemistry (9).
References:
| [1] | Oliver Plietzsch, Christine Inge Schilling, Mariyan Tolev, Martin Nieger, Clemens Richert, Thierry Muller, Stefan Bräse, Four-Fold Click Reactions: Generation of Tetrahedral Methane- and Adamantane-Based Building Blocks for Higher Molecular Assemblies, Org. Biomol. Chem. 2009, 7, 4734–4743 |
| [2] | Christine Inge Schilling, Oliver Plietzsch, Thierry Muller, Stefan Bräse, Fourfold Suzuki-Miyaura and Sonogashira cross-coupling reaction on tetrahedral methane and adamantane derivatives, Eur. J. Org. Chem., 2011, 1743–1754 |
| [3] | Olivier Plietzsch, Christine Inge Schilling, Thierry Muller, Stefan Bräse, Asymmetric synthesis of chiral tectons with tetrapodal symmetry: Fourfold asymmetric reactions, Tetrahedron: Asymmetry 2010, 21, 1474–1479 |
| [4] | Philippe Pierrat, Sylvia Vanderheiden, Thierry Muller, Stefan Bräse, Functionalization of Hexakis Methanofullerene Malonate Crown-Ethers: Promising Octahedral Building Blocks for Molecular Networks, Chem. Commun. 2009, 1748–1750 |
| [5] | Martin Meng, Carolin Ahlborn, Matthias Bauer, Oliver Plietzsch, Sahhid A. Soomro, Arunoday Singh, Thierry Muller, Wolfgang Wenzel, Stefan Bräse, Clemens Richert, Two Base Pair Duplexes Suffice to Build a Novel Material, ChemBioChem 2009, 10, 1335–1339 |
| [6] | Arunoday Singh, Mariyan Tolev, Martin Meng, Konstantin Klenin, Oliver Plietzsch, Christine Inge Schilling, Thierry Muller, Martin Nieger, Stefan Bräse, Wolfgang Wenzel, Clemens Richert, Branched DNA that Forms a Solid at 95 °C, Angew. Chem. Int. Ed., 2011, 50, 3227–3231; Angew. Chem. 2011, 123, 3285–3289 |
| [7] | Weigang Lu, Daqiang Yuan, Dan Zhao, Christine Inge Schilling, Oliver Plietzsch, Thierry Muller, Stefan Bräse, Johannes Guenther, Janet Blümel, Rajamani Krishna, Zhen Li, and Hong-Cai Zhou, Porous Organic Frameworks: Porosity and Clean Energy Applications, Chem. Mat. 2010, 22, 5964–5972 |
| [8] | Andrew J. Inglis, Philippe Pierrat, Thierry Muller, Stefan Bräse, Christopher Barner-Kowollik, Well-Defined Star Shaped Polymer-Fullerene Hybrids via Click Chemistry, Soft Matter 2010, 6, 82–84 |
| [9] | Oliver Plietzsch, Christine I. Schilling, Tobias Grab, Stephan L. Grage, Anne S. Ulrich, Angiolina Comotti, Piero Sozzani, Thierry Muller, and Stefan Bräse, Click Chemistry Produces Hyper-Cross-linked Polymers with Tetrahedral Cores, New. J. Chem., accepted (NJ-LET-04-2011-020370) |
List of Publications 2006-2011 as PDF
Subproject Report 2006-2010 as PDF